 
The factor-label method yields the desired cancellation of units, and the computed result is on the order of 10 22 as expected. The lessons learned in this post will continue through the study of chemical reactions and equations.\] Generally, this number is the one with the decimal point. The table has mass, or molar mass, shown as the number in the bottom of each square. You can see that The molar mass of Sodium is 22.990 g/mol. 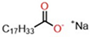
So let’s look at the molar mass of Sodium, Oxygen and Hydrogen from the above periodic table. 
Now in NaOH, there is 1 Sodium atom, 1 Oxygen atom and 1 Hydrogen atom. We also learned how to calculate the molar mass of a compound using the periodic table and how to convert mass into moles. Periodic Table with Mass Numbers and Atomic Symbols. You can see the molar mass value of all the atoms from this periodic table. However, if we have 6. For normal samples from earth with typical isotope composition, the atomic weight can be approximated by the standard atomic weight or the conventional atomic weight. According to the periodic table, the atomic mass of aluminum is 26.98 amu, copper is 63.55 amu, and carbon is 12.01 amu.Since 1 amu is only 1.674 × 10 24 g, these masses would be way too small to measure on ordinary laboratory equipment. Now here we have to find the molar mass of C3H8 (Propane). Because the molar mass of any molecule (or compound) can be calculated by simply adding the molar masses of individual atoms. We learned about Avogadro’s number and how it relates to the mole, which is a unit used to express the amount of a substance. The molar mass of atoms of an element is given by the relative atomic mass of the element multiplied by the molar mass constant, M u 1 × 10 3 kgmol 1. If you have a periodic table with you, then you can easily calculate the molar mass of C3H8 (Propane). 
According to the periodic table, 1 mol of U has a mass of 238.03 g, so the mass of 2 mol is twice that, or 476.06 g. In conclusion, understanding molar mass is an essential concept in chemistry as it allows us to relate the amount of a substance to the number of particles present in it. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
